Key Stats for Johnson & Johnson Stock
- Current Price: $230.42
- Target Price (Mid): ~$321
- Street Target: ~$253
- Potential Total Return: ~39%
- Annualized IRR: ~7% / year
- Earnings Reaction: (0.60%) April 14, 2026
- Max Drawdown: 10.96% May 8, 2026
Now Live: Discover how much upside your favorite stocks could have using TIKR’s new Valuation Model (It’s free) >>>
What Happened?
Johnson & Johnson (JNJ) reported Q1 2026 sales of $24.06 billion, up 9.9% year over year, and raised full-year guidance to approximately $100.8 billion. The stock slipped 0.60% on April 14. That reaction tells you the quarter was already expected.
What wasn’t in the earnings release was the pipeline roadmap John Reed, Executive Vice President of Pharma R&D, laid out on May 12 at the Bank of America Global Healthcare Conference. He covered where ICOTYDE goes after psoriasis, what the RYBREVANT data at ASCO looks like, and why SPRAVATO’s growth is more durable than the headline number suggests.
The core question in JNJ right now is whether ICOTYDE in IBD, RYBREVANT in head and neck cancer, and the DUET co-antibody therapeutic can build a second growth wave that sustains Innovative Medicine expansion into the early 2030s. Reed’s answers on May 12 are what this article unpacks.
ICOTYDE: Why the Label Matters as Much as the Drug
ICOTYDE (icotrokinra) received FDA approval in March 2026 for moderate-to-severe plaque psoriasis. It is an oral, once-daily peptide that blocks the IL-23 receptor, the same pathway as leading injectable biologics, and went 5 for 5 across its Phase III ICONIC program. The published 52-week data showed that up to around half of patients achieved completely clear skin, on par with the best biologics.
Reed’s commercial framing at BofA started with a structural gap: only 20% to 30% of patients eligible for a biologic in autoimmune disease are actually taking one. Most won’t commit to a lifetime of injections. ICOTYDE’s label removes the main friction point: no laboratory monitoring required, no TB testing upfront, and a dermatologist can write the prescription immediately.
The bigger opportunity is IBD (inflammatory bowel disease). Reed confirmed J&J is heading straight to Phase III in ulcerative colitis based on DUET dose-ranging data and is running a seamless Phase IIb/III in Crohn’s. Fewer than half of IBD patients achieve sustained remission on current therapies. If ICOTYDE delivers biologic-level efficacy in IBD Phase III, management has called it one of J&J’s potential largest products ever, sitting alongside TREMFYA, which is already targeting more than $10 billion in peak-year sales.

See historical and forward estimates for Johnson & Johnson stock (It’s free!) >>>
RYBREVANT: The ASCO Catalyst Two Weeks Away
RYBREVANT FASPRO (amivantamab) is a bispecific antibody, a drug engineered to block two cancer-driving proteins simultaneously, already approved in lung cancer, and now being studied in head and neck and colorectal cancer.
Head and neck squamous cell carcinoma is the eighth most common cancer globally. In second-line, the standard of care delivers response rates of just 10% to 20%. RYBREVANT received FDA Breakthrough Therapy Designation in February 2026 as second-line monotherapy in this setting, based on Phase 1b/2 OrigAMI-4 data showing rapid, durable responses in a heavily pretreated population.
The pivotal OrigAMI-4 second-line monotherapy data will be presented at ASCO 2026, running May 29 through June 2 in Chicago. Reed told BofA attendees that J&J believes these data could support a regulatory filing. That is the most important near-term catalyst for this stock.
J&J’s multiple myeloma franchise underscores what that kind of pipeline execution can look like. TECVAYLI, its first-in-class T-cell engager for myeloma, received its most recent second-line approval in just 55 days after submission, after the FDA voluntarily offered a Commissioner’s Priority Review Voucher. A second Phase III study, MajesTEC-9, is also being presented at ASCO. Reed noted at BofA that J&J’s myeloma combinations are generating 100% MRD (minimal residual disease) negativity rates in pilot frontline studies, a result he described as approaching a cure.
SPRAVATO: What the 60% Growth Rate Doesn’t Tell You
SPRAVATO (esketamine) grew more than 60% in 2025. The mechanism behind that durability is what Reed explained at BofA. Unlike traditional antidepressants that adjust neurotransmitter levels and require weeks to work, SPRAVATO promotes neuroplasticity, the brain’s ability to form new neural connections through NMDA receptors, producing effects within hours.
Reed stated at BofA that 50% of patients remain in remission for five years on the drug. This figure has not been published in a peer-reviewed primary endpoint study; it reflects management’s characterization of long-term program data. He also noted that J&J is in ongoing discussions with regulators about moving SPRAVATO toward home administration, which would remove the current two-hour clinic monitoring requirement and expand the addressable population.
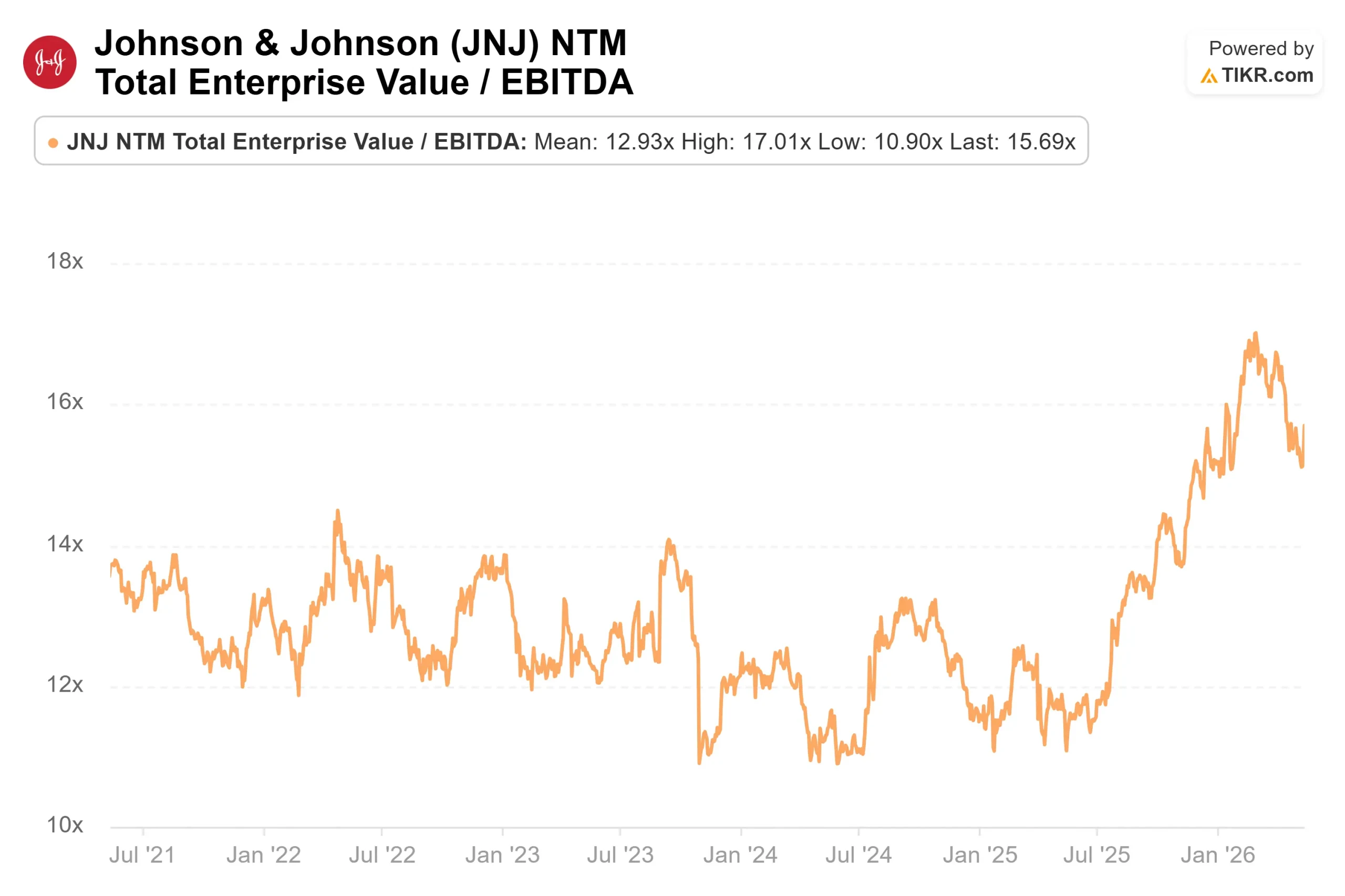
Among pharmaceutical peers tracked on TIKR’s Competitors page, JNJ trades at 15.69x NTM EV/EBITDA, compared to Roche at 10.73x, Novartis at 13.62x, and Pfizer at 8.18x. The premium reflects J&J’s active Phase III pipeline breadth and double-digit Innovative Medicine growth that peers are not currently matching. Whether it holds or expands depends on what ASCO delivers.
See how Visa Inc. performs against its peers in TIKR (It’s free!) >>>
TIKR Advanced Model Analysis
- Current Price: $230.42
- Target Price (Mid): ~$321
- Potential Total Return: ~39%
- Annualized IRR: ~7% / year

See analysts’ growth forecasts and price targets for Johnson & Johnson stock (It’s free!) >>>
The mid-case model assumes a revenue compound annual growth rate of approximately 7% through 2030, driven by Innovative Medicine scaling into new indications, ICOTYDE in IBD and psoriatic arthritis, RYBREVANT beyond lung cancer, and steady MedTech expansion. Net income profit margins are projected to expand toward approximately 31%, reflecting a mix shift as Innovative Medicine grows as a share of total revenue, particularly after the planned mid-2027 DePuy Synthes separation.
The upside case requires ICOTYDE to reach blockbuster scale in IBD and RYBREVANT to win a head-and-neck approval. The primary downside risk is talc litigation: Q1 2026 alone included approximately $0.3 billion in talc-related charges, following a $7 billion reserve reversal that benefited the prior year. If reserves need a significant step-up, 2026 EPS faces compression.
The Street’s mean target of approximately $253 based on 10 Buys, 5 Outperforms, 9 Holds, 1 Underperform, and 1 Sell from 26 analysts has not been updated for the BofA pipeline commentary or the incoming ASCO data. A positive OrigAMI-4 readout is the most direct path to closing that gap.
Conclusion
Watch ASCO 2026, running May 29 to June 2. The OrigAMI-4 second-line monotherapy data for RYBREVANT in head and neck cancer is the single catalyst that will either expand or constrain this valuation story. Reed told the BofA audience the data could support a regulatory filing, in a disease where the current standard of care delivers 10% to 20% response rates.
A result well above that bar, with durable responses, confirms a filing and forces the Street to reprice RYBREVANT’s contribution. A weak result leaves the stock reliant on the existing portfolio ramp alone. The data will be available in two weeks. The Street’s ~$253 target is already priced for the existing business. Everything above that depends on what ASCO shows.
See what stocks billionaire investors are buying so you can follow the smart money with TIKR.
Should You Invest in Johnson & Johnson?
The only way to really know is to look at the numbers yourself. TIKR gives you free access to the same institutional-quality financial data that professional analysts use to answer exactly that question.
Pull up Johnson & Johnson, and you’ll see years of historical financials, what Wall Street analysts expect for revenue and earnings in the quarters ahead, how valuation multiples have moved over time, and whether price targets are trending up or down.
You can build a free watchlist to track Johnson & Johnson alongside every other stock on your radar. No credit card required. Just the data you need to decide for yourself.
Analyze Johnson & Johnson on TIKR Free →
Looking for New Opportunities?
- See what stocks billionaire investors are buying so you can follow the smart money.
- Analyze stocks in as little as 5 minutes with TIKR’s all-in-one, easy-to-use platform.
- The more rocks you overturn… the more opportunities you’ll uncover. Search 100K+ global stocks, global top investor holdings, and more with TIKR.
Disclaimer:
Please note that the articles on TIKR are not intended to serve as investment or financial advice from TIKR or our content team, nor are they recommendations to buy or sell any stocks. We create our content based on TIKR Terminal’s investment data and analysts’ estimates. Our analysis might not include recent company news or important updates. TIKR has no position in any stocks mentioned. Thank you for reading, and happy investing!







